 
We have different bond strengths and reactivities in several existing molecules. We have different types of chemical bonds: ionic, covalent, metallic, hydrogen, and so on. If we get to learn why certain atoms come close and combine to form complex chemical structures and the different types of bonds that they form to achieve this complexity and formation of bigger compounds is to understand the chemical bonding of any given molecule or ion. The key to learning any compound thoroughly from the core is via its bonding nature and strength. The vast branches of usage do not negate the fact that BrF3 is highly reactive in water and quite toxic resulting in different human illnesses ranging from skin burns to eye ulcers and irritation in the respiratory system. 
Not only this, it has its application as an oxidizer in rocket propellants, as well as, acts as powerful inorganic solvents and even in manufacturing of uranium combined halogens for nuclear fuels. Highly soluble in sulfuric acid and acting as fluorine donors are some of the properties of bromine trifluoride. Also, this bearing both Br and F can give rise to HBr and HF acids. 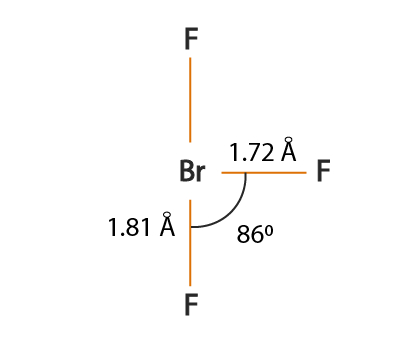
Since BrF3 consists of three fluorine molecules, this is a strong agent of fluorination. Here’s how we can synthesize or prepare the compound BrF3 : Having a straw i.e, colorless to yellow appearance, this chemical compound has several applications but also comes with a number of limitations and hazard issues. BrF3, known as Bromine Trifluoride, is a fuming liquid consisting of inter-halogen combinations and bearing a pungent smell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
